Anion Exchange Membranes
Ionic Liquids
Battery Electrolytes
Ionic liquids are salts in the liquid state that serve as highly stable and non-volatile electrolytes for electrochemical systems. They offer excellent ionic conductivity, wide electrochemical windows, and enhanced thermal stability. These properties make them ideal for applications in batteries, supercapacitors, and green energy technologies.
Zinc battery electrolytes, often paired with antisolvents, are engineered to enhance stability, suppress dendrite formation, and improve cycling performance. Antisolvents help regulate solvation structure and reduce unwanted side reactions, leading to higher efficiency and longer battery life. These advancements support the development of safe, cost-effective, and sustainable energy storage systems.
Anion Exchange Membranes (AEMs) are advanced polymer materials that selectively transport negatively charged ions while blocking others, enabling efficient electrochemical processes. They play a critical role in technologies such as fuel cells, water electrolysis, and energy storage systems. AEMs offer a promising pathway toward cost-effective and sustainable clean energy solutions.
Softwares: Gromacs, CP2K, Gaussian 16 and LAMMPS, VMD, Packmol, Molden etc.
Google Scholar Profile: Link
ORCID: https://orcid.org/0000-0001-9186-1970
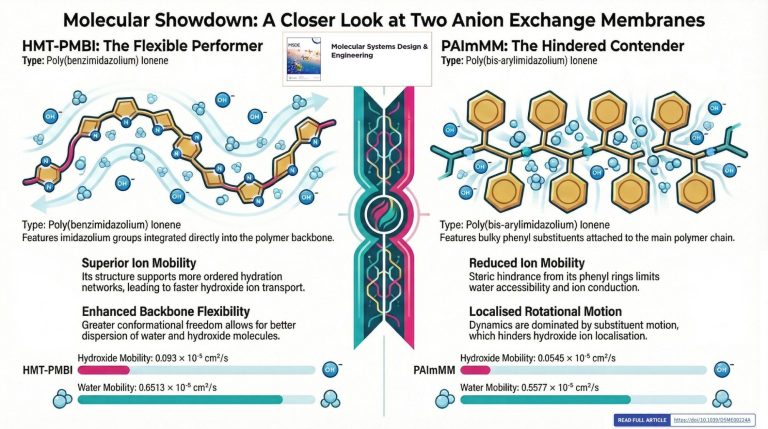
Atomistic insights into structure–morphology relationships in hydrated poly(benzimidazolium) and poly(bis-arylimidazolium) ionene membranes
S. Dagar, A. P. Sunda*
Mol. Syst. Des. Eng., 2026, 11, 255-268
https://doi.org/10.1039/D5ME00224A
Anion exchange membranes (AEMs) represent a rapidly emerging field due to their ability to facilitate efficient hydroxide (OH−) ion transport, offering a more environmentally friendly alternative to traditional proton exchange membranes (PEMs). One of the key factors influencing AEM performance is the choice of membrane material and functional groups.
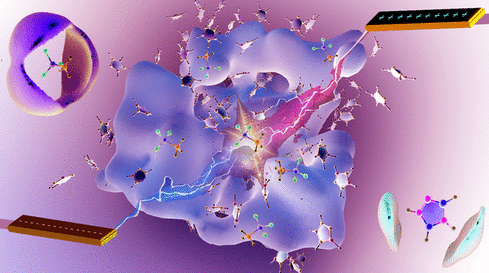
Molecular Insights into Binary Ionic Melts of Protic Ionic Liquid 1,2,4-Triazolium Methanesulfonate and Methanesulfonic Acid Electrolytes
S. Dagar, S. Liu, J. Luo*, A. P. Sunda*
J. Phys. Chem. B 2025, 129, 1, 338–347
https://doi.org/10.1021/acs.jpcb.4c05867
Binary ionic melts formed by a protic ionic liquid (PIL) 1,2,4-triazolium methanesulfonate ([TAZ][MS]) dissolved in methanesulfonic acid are studied as non-stoichiometric electrolytes. The composition-driven structure–property relationship of methanesulfonic acid is explored at varying molar fraction ratios from 0/100 to 10/90, 20/80, and 30/70 by the addition of 1,2,4-triazolium methanesulfonate [TAZ][MS] IL.
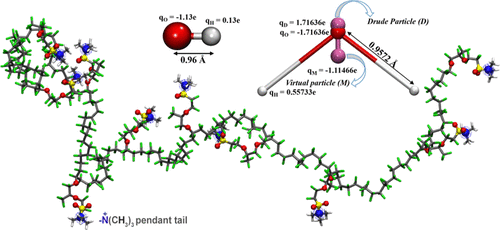
Molecular Insights of Hydrated Perfluoro Quaternary Ammonium Anion-Exchange Membranes: Ionomer Structure and Hydroxide Ion Transport
P. Sharma, A. P. Sunda*
ACS Appl. Polym. Mater. 2025, 7, 3, 1886–1895
https://doi.org/10.1021/acsapm.4c03696
Perfluoro membranes are recognized electrolytes for fuel cell applications due to their outstanding electrochemical properties. Perfluoro membrane precursors are used to synthesize anion-exchange membranes with certain modifications. Quaternary ammonium-modified perfluoro membranes successfully demonstrated their suitability and performance for prolonged fuel cell operation.
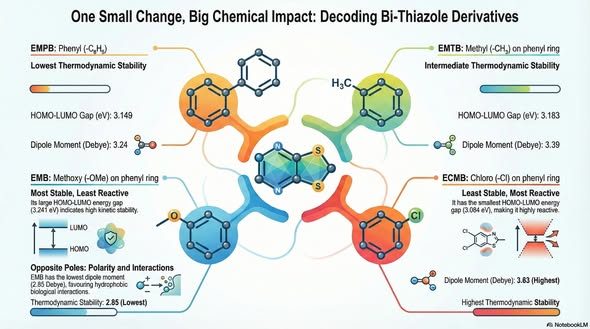
Molecular analysis of vibrational and structural characteristics in bi-thiazole derivatives
Dagar et al.
Discover Chemistry, 2026, 3, 1
https://rdcu.be/eXnIZ
Thiazole and bi-thiazole-based compounds’ vital role in broad-spectrum biologicalactivities attracts immeasurable research interest to investigate their molecularcharacteristics. This work aims to explore the molecular insights of bi-thiazolecompound ethyl 2’-(4-methoxyphenyl)-4’-methyl-2-phenyl-[4,5’-bithizole]-5-carboxylate (EMPB) and its methyl, methoxy and chloro derivatives such as EMTB,EMB, ECMB. B3LYP hybrid functional at the 6-31+G(d,p) basis set level theoryimplemented to get molecular geometry features, charge density distributionand insights into physicochemical properties.
SPONSORED/RESEARCH PROJECTS
- SERB Core Research Grant: F. CRG/2022/001938 (Completed)
Project Title: Microphase Separation .... Membrane Fuel Cell Applications Grant: INR 32,14,464/-
Awarded by: SERB (Now ANRF) New Delhi, Govt of India
Duration: 3-Years w.e.f. Jan 7, 2023
- UGC BSR Start-up Grant: F. 30/589-2021 (Completed)
Project Title: Polymeric Form of Ionic Liquids Grant: INR 10,00,000/-
Awarded by: University Grants Commission
Duration: 2-Years w.e.f. June 17, 2022
- Research Seed Money Award (Completed)
Project Title: Polymer Composite of ionic Liquids Grant: INR 2,00,000/-
Awarded by: J. C. Bose University of Science and Technology, YMCA
Duration: July 2021 to March 2023 (2-Years)
- DST INSPIRE Faculty Research Grant: DST/INSPIRE/04/2014/015731 [IFA14-MS31] (Completed)
Project Title: Ab Initio Molecular Dynamics simulation of Ionic Liquid doped Polymer Electrolyte Membranes and Platinum
Electrode Interface
Awarded by: Department of Science and Technology, Delhi
Duration: March 2015 to March 2020 (5-Years) Grant: INR 35,00,000/-
2nd Year Review Grading: Very Good
- Rajat Jayanti Vigyan Sancharak Fellowship: CO/S/TR/F09/2012 (Completed)
Project Title: Molecular Modeling in Design and Development of Novel Materials for Renewable Energy
Awarded by: National Council of Science & Technology, DST, New-Delhi.
Duration: Jan 2013 to Dec 2013 (1-Year) Grant: INR 2,29,000/-
Host Institute: Indian Institute of Science Education and Research (IISER), Pune